China has tightened the quality control on anti-malaria drugs sold to African countries with a newly-issued regulation and other efforts, said the State Food and Drug Administration (SFDA) here Monday.
According to the new regulation, China will only export anti-malaria drugs produced through a group of government-appointed pharmacy companies to African clients and carefully examine their products before export, said Wu Zhen, the SFDA deputy Director, at a press conference.
"We promise to sell safe and effective anti-malaria tablets to Africa," Wu said, adding that they did receive reports of sub-standard Chinese drugs sold in Africa.
Chinese researchers extracted a fast-acting, anti-malaria agent named artemisinin from a traditional Chinese herb in the 1970s. The tablets based on this subject are taking the majority of drug exports from China to Africa where about 3 million people die of malaria every year.
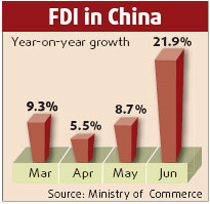
China's pharmacy industry, with an annual output value of 500 billion yuan (66.67 billion U.S. dollars), is under criticism for quality problems.
The problems turn up as the export volume increases. Also, part of the problem is that quality management in China and foreign countries is done differently, Wu said.
"Condemnation and complaint won't help to close loopholes in quality control of medicines. The most effective way is cooperation between China and export markets," he said.
He suggested that the two sides share information and technology improvements and, even better, develop a mechanism to jointly cope with the issue.
By October, China closed 300 drug and medical device manufacturers in a national campaign to regulate its pharmacy industry, which started last July.
(Xinhua News Agency December 4, 2007)